Management Review – A Must For Organizations
Management Responsibility
The other challenge has gotten to do with the caliber of people that participate in management reviews. Again, there are several instances where quality reviews are relegated to line managers whilst senior management excuse themselves to “strategic planning”. It is not for nothing that the ISO Standard recommends management review to comprise of top management, a quality management representative who chairs the meeting, line managers and compliance managers. It is also important to invite any other person who may give quality input into the review. The importance top management gives to quality and related issues in a business determines how far the company will thrive. Short term goals of seeing profits go up may give a company a lifeline for a few years, but a long-term plan that embraces the right management of quality gives a business oxygen for life.
Management Review Inputs
The first input will consider the status of actions from previous management reviews. This follows a typical review of actions in meetings. It is not prudent to keep adding actions at meetings without first considering previous actions and the status of those actions. Accountability is key to improvement. We laud achievements but sound reason should form the basis why we miss targets. Another input to management review is changes in external and internal issues that are relevant to the quality management system. In the course of the year, certain changes such as reviews in formulation, processes, external supply chain, regulatory issues, and a lot more may emerge.
These internal and external issues may have a direct impact on the quality management system such that, things should be done differently to accommodate them. The overall impact on quality of products and services should be positive and ultimately satisfy customer requirements. The third input to management review is information on the performance of the quality management system, including trends and indicators for: Nonconformities and corrective actions; Monitoring and measurement of results; Audit results, Customer feedback; Supplier and external provider issues; and Process performance and product conformity. All these pieces of information come as a result of active participation of individuals within the organization because these are mainly driven by records, trends and trend analysis and a cycle of activities geared towards closing the loop on nonconformities. Activities such as process audits, hygiene audits, supplier audits, consumer/customer complaints monitoring, just to mention a few are so crucial to bringing to the fore how well or otherwise an organization is performing with respect to quality management. The last input to management review is opportunities for continual improvement. This is usually ascertained from the overall performance of the system; what went well and could be improved and what didn’t go well and must be worked on.
Management Reviews Output
Conclusion
Related Stories
Product Packaging – Your Best Salesman!
Product Packaging - Your Best Salesman!“Competition is a great thing and critically important in any industry. I respect the companies that build their brand through innovation/great product, packaging, sharp marketing and clever ideas” John Robinson. A great product...
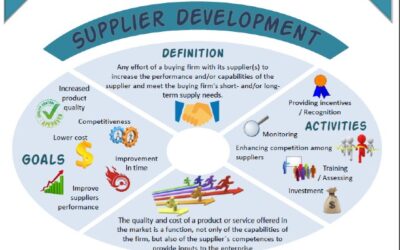
Supplier Development in a Post-Covid Era
Supplier Development in a Post-Covid EraI have used a greater part of my time last week in discussing world class manufacturing (WCM) with some trainees. Most of the lessons learned can actually be applied to the service industry. One definition pitches WCM as a...
Quality Circles III
The Quality Circles (Part 3)This will be the last in the series for Quality circles. This article would be looking at the core functions of the various layers in the structure of Quality Circles. The importanceof kaizen projects cannot be underestimated and Quality...
Get In Touch
HEAD OFFICE
Aviation Highway , East Airport ( Spintex)
Landmark: Endpoint Homeopathic clinic